The French Multiple Sclerosis Observatory

Note: Further information can be found in the French-language section of this site.
Observatoire Français de la Sclérose en Plaques (in French)The adoption of the EDMUS software by the majority of neurology departments specializing in Multiple Sclerosis (MS) in France allows the pooling of anonymized data on more than 30.000 French patients suffering from this disease, whose files are stored in the EDMUS format. This represents approximately half of the French patients, and almost all of those who are followed by neurologists, since in advanced stages of the disease patients are mostly cared for in long-stay homes. France is the first country in the world to benefit from such a source of information about the disease.
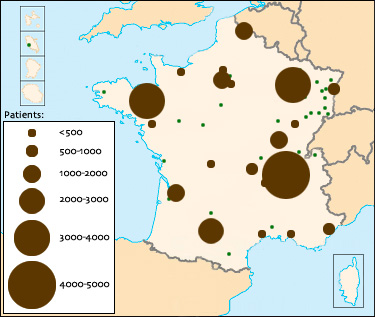
saved in the EDMUS format in France (in 2008)
Since its creation in 2003, the Observatory has been providing a "snapshot" of MS at the national level, which is updated and refined from year to year. This should help better understand the personal, professional and social impact of the disease, the effects of disease-modifying treatments, and the requirements for the management of the disease in France. The Observatory is ideally suited to epidemiological studies on MS at the national level.
This project has been conducted since 2003 under the sponsorship of the French Society of Neurology and of the States General of MS (national conference on MS), held in Paris on April 28, 2006 in the presence of the Minister of Health and Solidarity, Mr. Xavier Bertrand.
The OFSEP project selected among the Cohort projects of the
"Investments for the Future" program
The "Observatoire Français de la Sclérose en Plaques" (French Observatory of Multiple Sclerosis) (OFSEP) project, submitted in response to the Cohorts ANR tender of the "Investments for the Future" program, was selected by the Ministry of Higher Education and Research among 10 national projects selected from 44 submitted projects. The funding allocated amounts to more than 10 million euros.
With this funding, the OFSEP will be able to increase the size of the national cohort, approaching exhaustiveness, and to provide a real-time photograph of the disease in France. It will also be able to extend its scope of investigations, combining clinical data with biological samples and - a major advance - with imaging data. This selection should also enable the project to strengthen its collaboration with AFSSAPS (the French Medicines Agency) for systematic pharmaco-epidemiological studies within the framework of risk management plans, to develop its cooperation with the French Health Insurance System for medico-economic studies, and to establish a collaboration with the High Authority for Health, especially its Transparency Commission, to assess treatment efficacy in real life, beyond clinical trials.
By supporting OFSEP, the Ministry provides the country with a powerful epidemiological tool and an "MS plan" which are unique in the world.